121 S. Wilke Road, Suite 301, Arlington Heights, IL 60005
Home and Hospital Visits for Your Convenience
Serving Clients Across 7 Illinois Locations
Recent Blog Posts
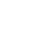
What can Happen When Surgical Equipment is Left Inside Your Body
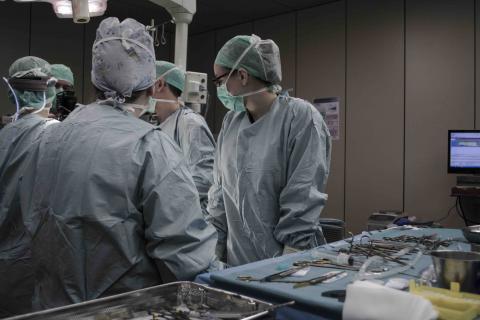
Being under anesthesia to receive surgery puts you in a vulnerable position. You have to trust your doctor and all the other healthcare professionals in the room to perform the surgery correctly and to have your best interest in mind. Usually, this is exactly what happens, and the patient makes a full recovery.
Sometimes, though, healthcare professionals make errors. One particularly dangerous type of surgical error is leaving surgical equipment inside the patient's body after surgery, which puts the patient at risk of fatal injury. Victims and in fatal cases, their loved ones, can pursue compensation related to surgical error damages through medical malpractice claims. If surgical equipment is left inside your body after surgery, you can experience one or more of the following:
You can Experience Chronic Pain
There is no other way to put it – having a foreign object in your body can be painful. Often, chronic pain after surgery is a sign that something is not right. It is normal to experience some lingering soreness or inflammation after surgery, but when pain is persistent, stinging, or shooting, the patient could have an object at the surgical site that should not be there.
Birth Control Recalled Due to Packaging Error

In late May 2018, pharmaceutical manufacturer Allergan voluntarily recalled approximately 170,000 sample packs of its Taytulla birth control pills due to a packaging error. In the recalled packs, lot 5620706 with a May 2019 expiration date, had their placebo pills at the beginning of the packs, rather than the end where they are supposed to be. These four placebo pills contain no hormones and are meant to give users a "break," during which they experience period-like bleeding, between three-week rounds of pills that contain hormones.
An incorrectly packaged medical product, such as the recalled birth control packages, is a defective medical product that can put patients at risk of suffering the effects of not receiving the medical care they need.
Product Manufacturers are Liable for the Damages Related to Their Products
What Evidence can I Use to Support My Car Accident Claim?

When you are injured in a collision with a negligent driver, you have the right to pursue monetary compensation for your damages through a personal injury claim. Just like pursuing compensation for your damages after any other type of accident, you need to use evidence to support your automobile accident claim. Your lawyer can help you obtain some of the evidence you need, but other pieces need to come directly from the scene of the accident. When you file a car accident claim, you need the following types of evidence to support your claim:
Evidence From the Scene of the Accident
In the moments immediately after the collision, you need to gather a few key pieces of evidence that will demonstrate:
- That a collision occurred;
- That you and the other driver were the parties involved in the collision; and
What Kinds of Damages can My Auto Accident Compensation Cover?

When you are involved in a car accident, you can be injured. Sometimes, car accident injuries are fairly minor, such as scratches and bruising. In other cases, they are severe, sometimes to the point of causing the victim to suffer a long-term or even permanent disability or disfigurement. When a victim is injured in a car accident because of another party's negligence, he or she may pursue compensation for related damages through a personal injury claim. Through this type of claim, the victim may pursue compensation for the following:
Medical Expenses
Medical bills for an injury sustained in a car accident can be steep. In some cases, a car accident victim needs extensive medical care after an accident and continuing care for months or even years afterward. In your personal injury claim, you can seek compensation for all of your medical expenses.
Lost Wages
Often, an injury requires the victim to take time off work to recover. When this happens and he or she misses the opportunity to earn wages, the victim may seek compensation for these lost wages in his or her personal injury claim.
Slip and Fall Hazards Lurking in Your Home

You can slip, fall, and suffer an injury anywhere. When a slip or fall occurs because of a property owner's negligence, such as a store owner who failed to promptly clean up a liquid spill, the property owner is liable for the victim's related damages. But not all slips and falls happen in public places; you can slip and fall in your own home. Below are a few common slip and fall hazards in American households. Take care to repair these hazards in your home to protect yourself and your loved ones from falling, especially if an elderly relative lives with you. Slips and falls, especially in the bathroom, are one of the top causes of injuries among elderly people.
Unsecured Wires
Sometimes, it is necessary to run a wire across a room to power an electronic device. When you have to do this, either run the wire along the wall to keep it out of any walking paths or if this is not possible, secure it to the floor with tape. Even a small wire can be a tripping hazard, especially in dimly lit rooms.
Slip, Trip and Fall Hazards You can Face in Public

A slip or fall can happen anywhere. When an individual is injured in a slip or fall on private property, the property's owner is liable for the victim's damages. This concept is known as premises liability. Premises liability covers all injuries caused by hazards the property owner knew about or should have known about. Failing to clean up a spill or take a similar action to mitigate a slip and fall hazard is an act of negligence on a property owner's part. Below are a few common causes of slips and falls in public places.
Wet Floors
A spill or puddle on a floor can be a dangerous slip and fall hazard, especially if the floor material beneath is slick tile. In certain retail settings, detergents and other liquid soaps can spill and cause an especially dangerous slip and fall hazard. A few public settings where wet floors can be prevalent are:
Johnson & Johnson Ordered to Pay $35 Million in Damages to Victims Who Suffered from Using Pelvic Mesh
On March 8, 2018, an Indianapolis federal court ruled that Johnson & Johnson and Ethicon, its medical device division, were negligent in the design and warnings about its Prolift pelvic mesh and therefore, liable for the damages the plaintiff, a 69-year-old woman who had the mesh implanted in 2009, suffered as a result of using the mesh. The court ordered the company to pay $35 million in compensation, $25 million of which were punitive damages.
This was not the first defective medical device lawsuit Johnson & Johnson faced related to the Prolift pelvic mesh. Courts in Pennsylvania and New Jersey have also ruled in favor of plaintiffs injured by the Prolift mesh in other lawsuits.
Complications of Pelvic Meshes
Pelvic meshes, also known as transvaginal meshes, are medical devices that are surgically implanted in the pelvis to control the effects of pelvic organ prolapse (POP) and stress urinary incontinence (SUI). Although this device is meant to improve the patient's quality of life by providing support for weakened pelvic floor muscles, it can actually put the patient at risk of experiencing the following:
HeartStart MRx Defibrillator Recalled for Defective Part

The United States Food and Drug Administration (FDA) recently issued a recall for the Philips HeartStart MRx defibrillator because it contains a faulty part that can put patients at risk of serious injury and death.
The defective medical device was recalled after it was determined that a glass tube inside the defibrillator has tiny cracks that allow gases to escape and cause the device to malfunction. These cracks also allow electrical surges to cross the defibrillator's resistors, potentially damaging them and causing the device to fail while it is in automated external defibrillator (AED) mode. When the device fails in this mode, it can still be operated manually. Healthcare providers who have the defibrillator are urged to have the defective glass tube repaired to prevent patient injuries and deaths. In their uncorrected state, these defibrillators can fail at any time.
What Should I do if My Medication is Recalled?

When a drug on the market is determined to be dangerous in some way, it may be recalled. This can be because the drug was not advertised properly, because a specific batch or all of the drug available is contaminated or compromised, new side effects are discovered, or because the drug in the packages on the market is not the drug labeled on the packages. If a drug you currently take is recalled, stop taking it and contact your healthcare provider or local pharmacy for further instruction. Recalls exist to remove potentially harmful products from the market.
A defective drug claim is a type of personal injury claim. Through this type of claim, a victim can seek compensation for his or her medical expenses, lost wages, and pain and suffering damages related to the use of a harmful drug. If you were given a drug after it was recalled or were harmed by taking a dangerous drug that should have been recalled, you could have grounds to file this type of claim.
Who can be Liable for Defective Medical Device Damages
When a patient suffers an injury because of a defective medical device, he or she can seek compensation for his or her related damages through a defective medical device claim. In order to file a successful claim, the patient must first determine which party is liable for his or her damages. This depends on the actual circumstances of the patient's case. If you were injured in an incident involving a faulty or defective medical device, discuss your case with an experienced personal injury lawyer to determine who is liable and how to pursue your claim.
The Device's Manufacturer
In many cases, a defective device's manufacturer is the party liable for the victim's damages. This can be because the device had a design defect, rendering it inherently unsafe, a manufacturing defect, which means the device was not manufactured according to its design and this lapse rendered the final product unsafe for users, or a marketing defect, which means the manufacturer did not disclose the device's potential hazards to physicians, patients, and healthcare providers who might purchase and use the device.

 Spanish
Spanish